1. Introduction
2. Experimental
2.1 Materials
2.2 Preparation of GAP-based binders
3. Results and Discussion
3.1 GAP-based networks cured under single curing system
3.2 GAP-based networks cured under dual (hetero-) curing system
4. Conclusion
1. Introduction
Since the 1970s, solid composite propellants have undergone significant advancements and have evolved into the contemporary propulsion systems widely used in launch vehicles for versatile satellites. This remarkable progress in both military and civilian space applications would not have been possible without the synergistic combination of hydroxyl terminated polybutadiene (HTPB) and ammonium perchlorate (AP) as a hydrocarbon binder and an oxidizer, respectively. Especially, HTPB - originally developed for industrial purposes such as rubber and tires- played a pivotal role in accelerating the adaptation of solid propellants for rockets and missile technologies thanks to its conjugated diene structure, enabling partial electron delocalization along the backbone which results in a mildly energetic character, while maintaining superior mechanical flexibility and toughness [1,2]. In addition, its relatively high functionality facilitates the formation of a densely crosslinked polymeric network, in accordance with the Flory-Stockmayer theory shown in Eq. (1), (2) and (3), further enhancing the structural integrity of the binder matrix [3,4].
for Homofunctional system
for Bifunctional system
for General gelation condition
P or Pc : Extent of reaction or critical conversion, f : Functionality, fA, fB : Functionality of monomer A and B, R : Stoichiometric ratio of functional groups
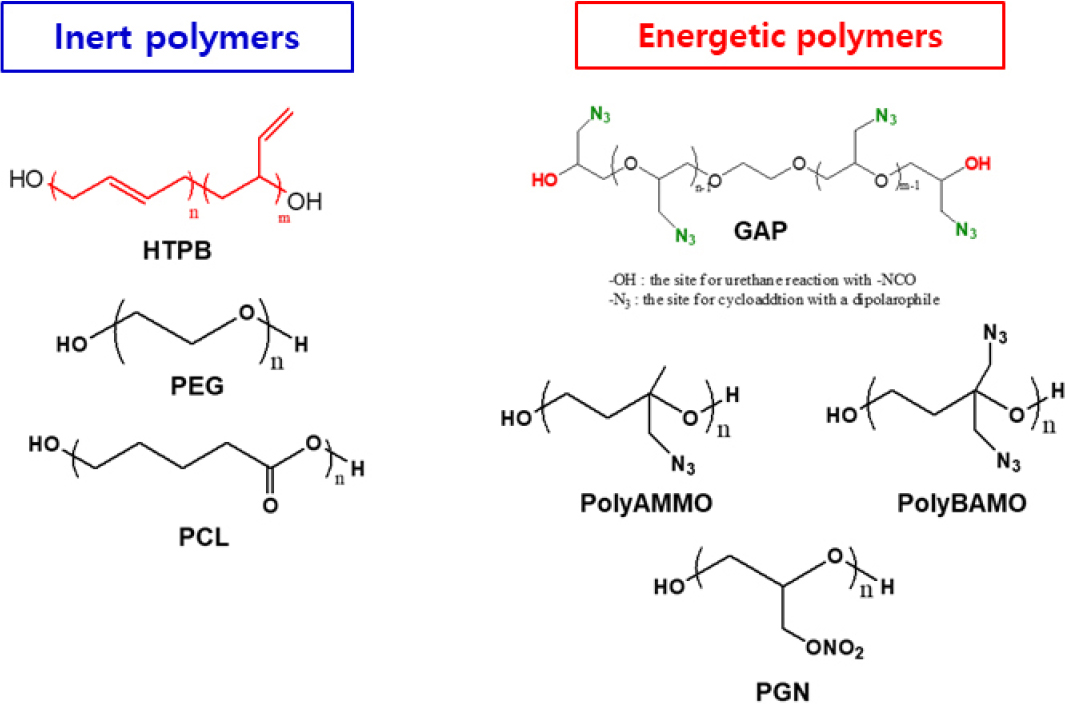
Although HTPB has contributed considerably to the development of composite solid propellants due to its excellent mechanical properties, positive heat of formation, and favorable processability, the performance of HTPB-based propellants - particularly in terms of specific impulse - has reached a practical plateau. To overcome this limitation, extensive research has been devoted to the development of high-energy binders incorporating energetic functional groups such as nitroesters (-ONO2), nitramines (-NNO2), and azides (-N=N+=N-) as shown in Fig. 1. Among these energetic polymers, polymers containing nitroester groups such as polyglycidyl nitrate (PGN) suffer from poor structural stability due to the intramolecular cyclization involving the nitrate ester group and the epoxide ring, leading to the formation of unstable cyclic species that deteriorate the thermal and chemical stability of binder networks [5,6,7]. Similarly, nitramine-functionalized polymers remain challenging to apply as binders in propellant formulations due to their highly viscous and sticky nature at room temperature, elevated glass transition temperature, and the high sensitivity associated with the nitramine group [8,9,10].
As a result, growing attention has been consistently paid to azide-functionalized polymers as promising energetic binders, owing to their excellent thermal stability and, by extension, chemical stability. In particular, azide polymers based on oxetane monomers, such as poly (3-azidomethyl-3-methyl oxetane, AMMO) and poly(3,3’-bis-(azidomethyl) oxetane, BAMO) have shown potential for elevating the performance of solid propellants [11,12,13]. Despite their high azide content, AMMO- and BAMO-based polymers-typified by higher melting points and Tg (glass transition temperature) values- exhibit high viscosity and sticky behavior at room temperature, which significantly hinders cast-curing processes and results in brittle mechanical properties [14,15]. Consequently, their use has been largely limited to thermoplastic formulations [16,17,18]. Compared to other azide-functional polymers (BAMO and AMMO), glycidyl azide polymer (GAP) synthesized from the azidation of hydroxyl-terminated polyepichlorohydrin has garnered significantly more attention as a practical energetic binder thanks to its excellent processability, lower glass transition temperature, good compatibility with other propellant component, and balanced energy-thermal stability profile [19,20,21].
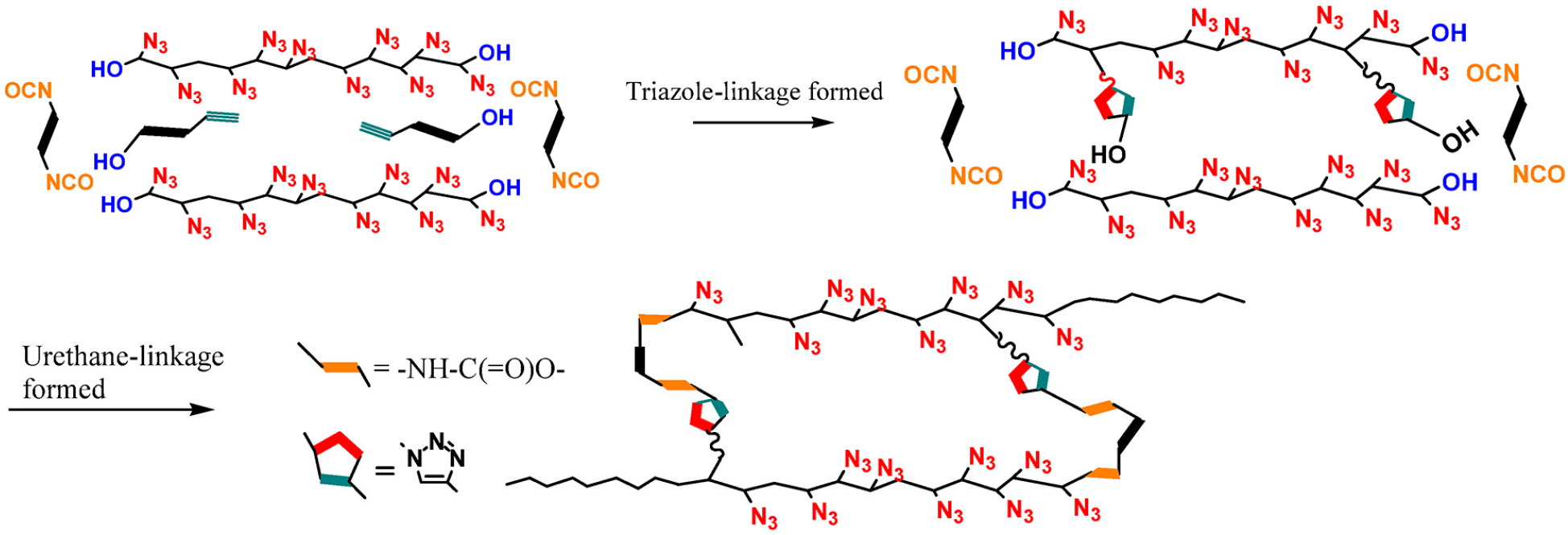
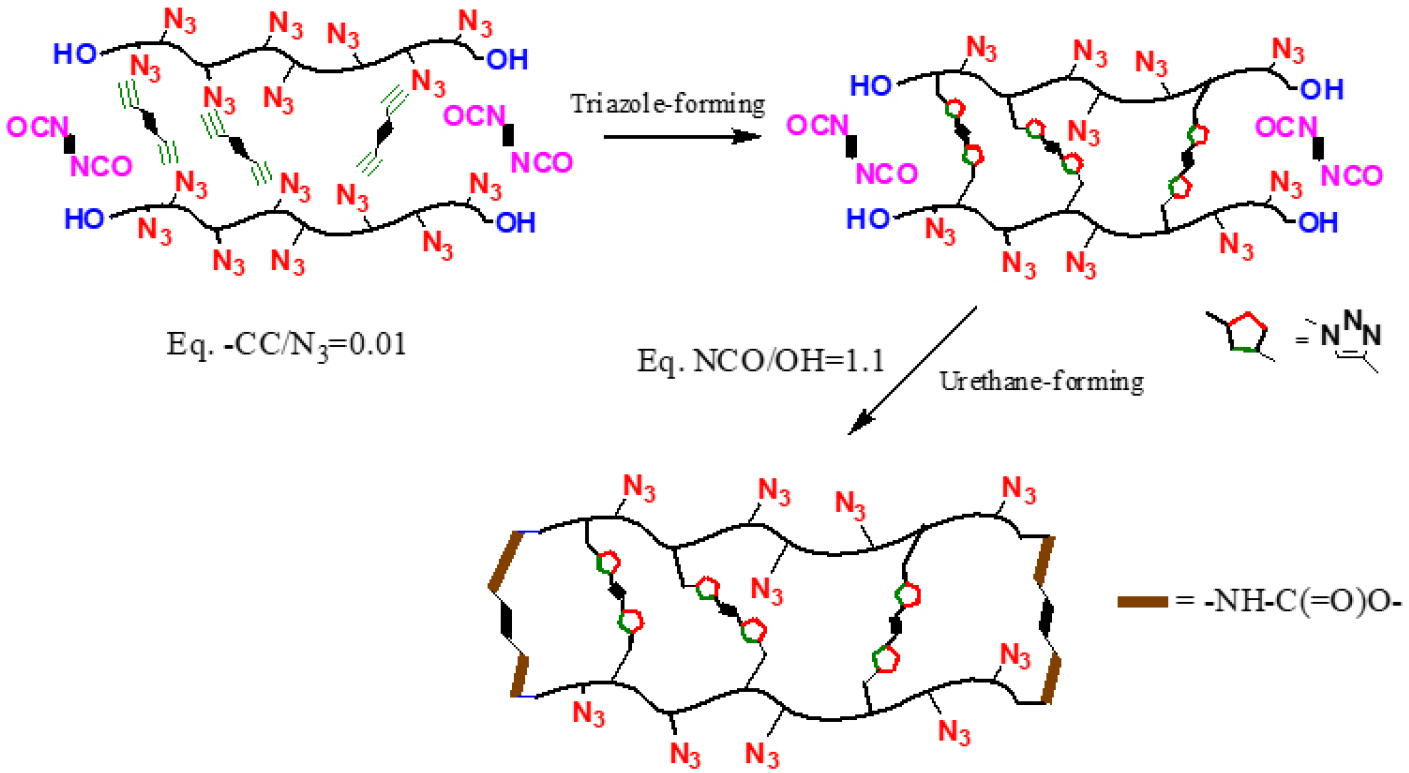
Despite its favorable energetic and processing characteristics, GAP suffers from inherent mechanical limitations due to the presence of methylene-linked azide pendant groups(-CH2-N3) in its main chain [22]. To overcome this issue, we have explored the incorporation of linear polymers such as polyethylene glycol (PEG) and polycaprolactone (PCL) to enhance flexibility and mechanical properties [23,24]. However, this strategy often leads to a reduction in the overall energy level of the binder system. To address this trade-off, we have developed a novel hetero-curing strategy to improve mechanical properties of GAP-based networks without compromising their energy density [25,26,27]. As illustrated in Fig. 1, GAP possesses terminal hydroxyl groups capable of reacting with isocyanates, while also featuring pendant azide groups that serve as key energetic moieties. These azide groups readily undergo 1,3-dipolar cycloaddition with dipolarphiles like alkenes (-C=C-) as well as alkynes (-C≡C-) [28,29].
In our approach, the terminal hydroxyl groups of GAP were first coupled with diisocyanates. However, as shown in Eq. (2) and (3), this alone does not lead to a crosslinked network. Therefore, to establish a three-dimensional network, we introduced di-alkyne compounds that can react with the pendant azide groups, thereby significantly strengthening the binder network. Given that azide groups can also react with alkene functionalities under similar conditions, we extended our investigation to include not only already-used alkyne such as bis-propagyl succinate (BPS) but also a series of novel alkyne- and alkene-based dipolarophiles. Moreover, we employed an asymmetric difunctional crosslinker bearing a hydroxyl group on one end and an alkyne on the other, enabling dual reactivity with GAP’s terminal and side-chain functional groups. Through this approach, we aimed to identify the optimal curing combination that would yield the best mechanical performance of the GAP-based binder system.
2. Experimental
2.1 Materials
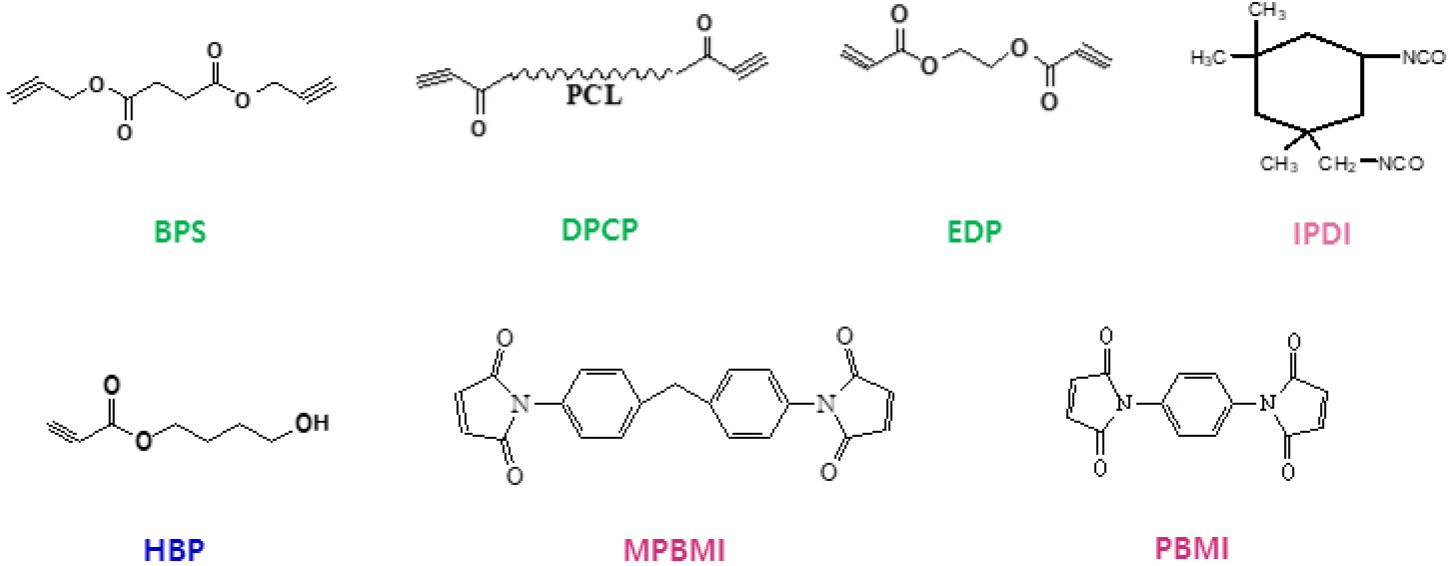
An azide polymer, GAP, was purchased from EURENCO under the trade name of PGA diol. Difunctional isocyanate compound for linking hydroxyl-terminated chain-ends of GAP, isophorone diisocyanate (IPDI) was obtained from Mobay. As a curing catalyst for urethane reaction, triphenyl bismuth (TPB) was acquired from Samchun Pure Chemical. Dialkyne-terminal curatives as a dipolarophile compound, BPS, ethlyene dipropiolate (EDP), and dipropiolate polycaprolactone (DPCL) as well as an unsymmetric difunctional curative, hydroxyl butylpropiolate (HBP) were synthesized as described in our earlier works [27,30]. Another dipolarophiles for the reaction with azides, dialkene-terminal compounds, maleic imide-type compounds (phenyl bismaleic imide (PBMI), methylene phenyl bismaleic imide (MPBMI)) were purchased from Aldrich co. All compounds applied for the formation of GAP-based networks are illustrated in Fig. 2.
2.2 Preparation of GAP-based binders
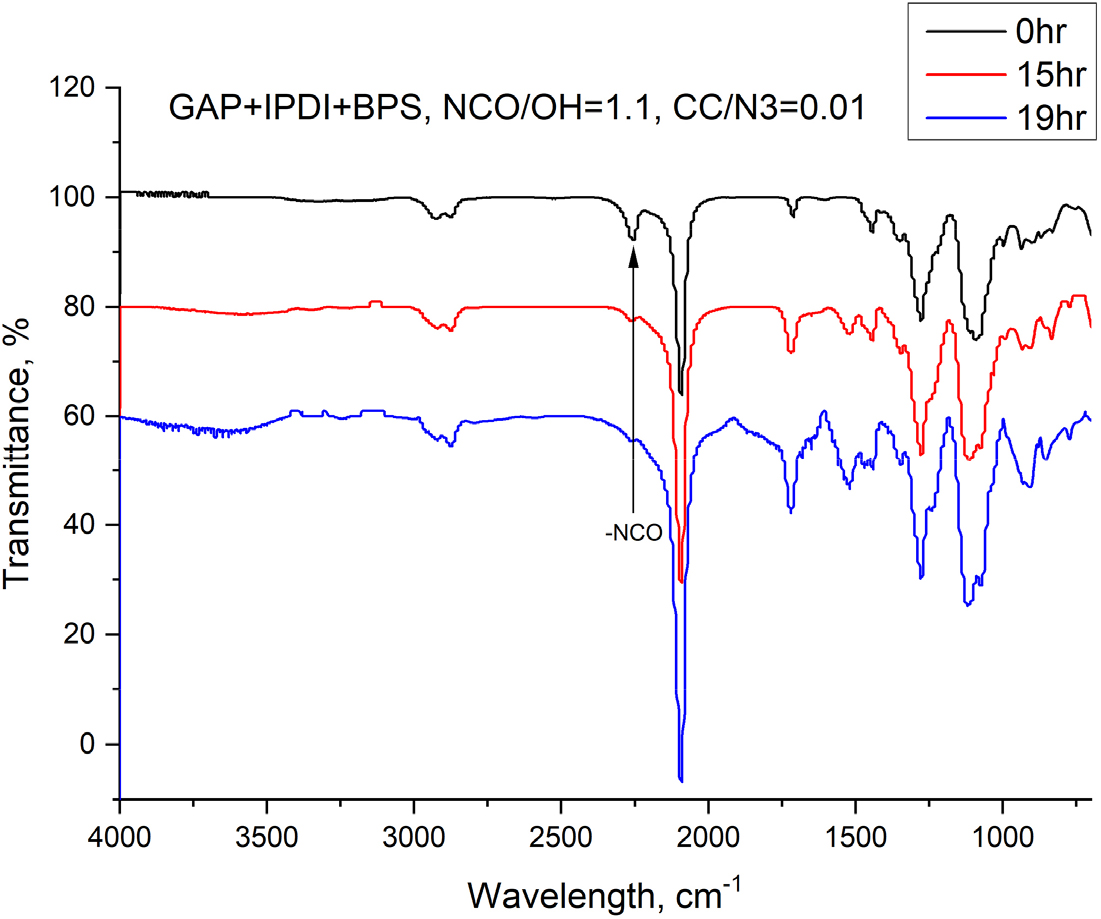
All of the reagents were dried overnight in a vacuum oven at 60°C before use. Under a hetero (or dual)-curing system composed of urethane moieties at the ends of GAP chains and triazole (or triazoline) moieties at the side-chains, the binders were prepared based on the equivalent ratio (-NCO/-OH) of 1.1 and 0.1 wt% of TPB was introduced, followed by adding the certain amount of a dipolarophile to form the three-dimensional networks. After well-stirring under vacuum, the freshly mixed formulations were poured in a mold and then, cured at 50°C for a week. All crosslinked networks were cut into dumbbell-shaped (33 × 4.0 × 3.5 mm) and were subject to the mechanical measurements using a universal testing machine (Model 5567, Instron, USA) at a crosshead rate of 50 mm/min at room temperature. The mechanical properties of GAP-based polymer networks were comparatively evaluated using a series of dipolarophiles with either alkyne or alkene functionality. To ensure a consistent basis for comparison, the amount of each dipolarophile was adjusted so that the molar ratio of dipolarophile functional group to azide groups remained consistent across all systems. Based on the average azide content GAP-approximately over 100 azide units per chain- the amount of each dipolarophile was fixed to deliver an equivalent ratio of 0.002 (alkyne, -C≡C (or alkene, -C=C)/azide, -N3). FT-IR spectral measurement using a Nocolet NEXUS model ATR system was carried out to monitor the urethane reaction between hydroxyl groups in GAP and isocyanate groups in IPDI.
3. Results and Discussion
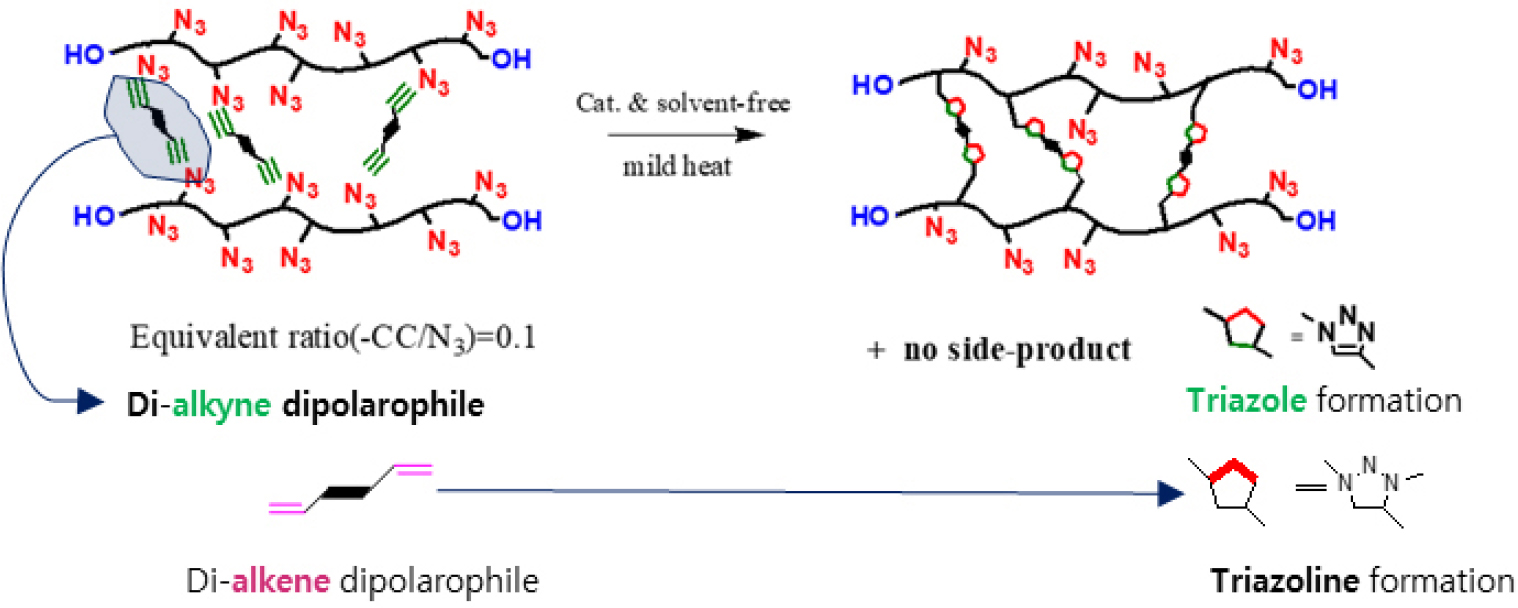
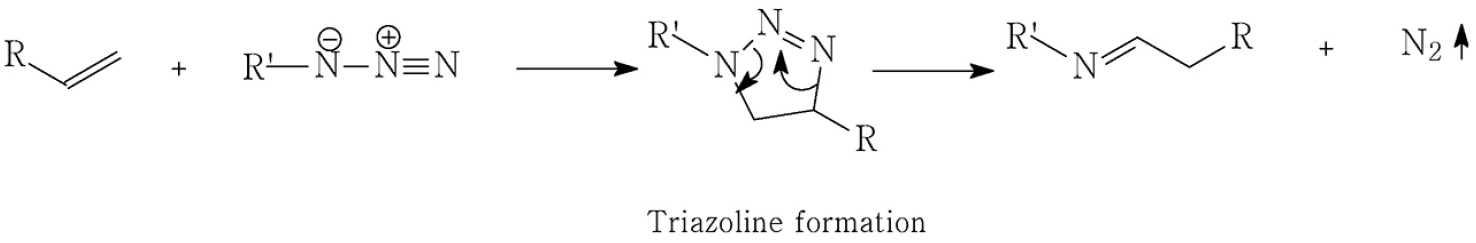
As previously introduced, GAP contains two distinct reactive sites in its polymer backbone: terminal hydroxyl groups and pendant azide moieties. The azide functionality, a typical 1,3-dipole, readily undergoes [3+2] cycloaddition with alkyne or alkenes to form triazole or triazoline rings, respectively. These cycloadditions-especially those involving alkynes-have been extensively studied as part of ‘Click chemistry’, owing to their regioselectivity and mild reaction conditions, often facilitated by copper(I) catalysis as shown in Scheme 1.
In our work, however, our objective was to form the crosslinked network without any kinds of catalysts or promotors such as copper. This was enabled by the high density of azide functionalities in GAP (approximately 100 repeat units per chain), which considerably elevates the local concentration of reactive sites, favorably influencing reaction kinetics in a non-catalyst environment. In addition, we employed alkyne and alkenes activated by nearby electron-withdrawing groups (e.g., esters, O-C(=O)- or carbonyls, -C(=O)-), which enhanced the dipolarophilic reactivity toward azides under mild, catalyst-free environment. Unlike triazoles, as illustrated in Scheme 2, triazolines formed via azide-alkene 1,3-dipolar cycloaddition are known to be structurally less stable due to their non-aromatic character, which makes them susceptible to thermal or chemical degradation [31]. To address this limitation, we employed alkene compounds bearing proximal electron-withdrawing groups like maleimide derivatives, which are known to stabilize the triazoline ring by delocalizing electron density and improving conformational rigidity [32]. This approach enabled the formation of a more robust and stable network. These 1,3-dipolar cycloaddition products-multinitrogen heterocycles-served as primary linkages in the GAP-based network.
Subsequently, the terminal hydroxyl groups of GAP were reacted with diisocyanate to introduce urethane links, forming a densely crosslinked dual (or hetero)-network structure as depicted in Scheme 3. Moreover, an asymmetric bifunctional crosslinker (HBP) containing both a hydroxyl-reactive isocyanate group and an azide-reactive alkyne moiety was also incorporated. These molecules enabled simultaneous or sequential covalent connections at both chain-ends and side chains, allowing for a more controlled and integrated polymer network architecture as shown in Scheme 4.
Based on this dual-network curing strategy, a series of GAP-based binders were synthesized using structurally diverse alkyne and alkenes. In the following sections, the mechanical performance of each binder system is evaluated, and the effects of different dipolarophiles on the resulting polymer network are comparatively discussed.
Scheme 3.
Formation of a dual (hetero-)crosslinking polymeric network via terminal hydroxyl-isocyanate reaction and pendant azide cycloaddition [27].
3.1 GAP-based networks cured under single curing system
Table 1 summarizes the mechanical properties of GAP-based polymer networks cured exclusively with the previously studied dialkyne compound, BPS. It was observed that a minimum of 7.0% of BPS was required to initiate effective network formation. Increasing the amount of 10.0% of BPS yielded a cured elastomer with sufficient hardness feasible for binder applications in solid propellants. However, as anticipated, the mechanical performance -particularly the strain at max. stress- was significantly limited, likely due to the presence of free dangling chains caused by side-chain-only crosslinking [25].
Based on these results, a maleimide-type alkene compound, PBMI, was selected as an alternative azide-reactive crosslinker and applied at the same 10.0% loading level. However, as illustrated in Fig. 3(a), solidified polymer network was not formed; the curing was incomplete, and the formulation exhibited a degree of foaming. This phenomenon is attributed to the thermal decomposition of triazoline rings formed during the azide-alkene cycloaddition reaction. As previously noted, triazolines are known to thermally unstable due to their non-aromatic nature, and prolonged exposure at the curing temperature (50°C for one week) likely triggered their degradation, releasing nitrogen gas.
Despite incorporation of a carbonyl-based electron-withdrawing group adjacent to the alkene moiety in maleimide - intended to stabilize the triazoline ring - this approach proved insufficient under the current curing environment. These findings suggest that only triazoline-based crosslinking, while chemically plausible, may pose limitations in practical thermal curing applications unless further stabilization strategies are introduced.
Table 1.
Mechanical characteristics of single (homo-) triazole-cured GAP-based networks (BPS only used as an alkyne linker).
| NO. | Alkyne linkers | Stress at max. strain, MPa | Strain at max. stress, % |
Modulus, MPa |
Hardness, Shore A, Hs | |
| GAP 1 | BPS | 5% | Uncured | |||
| GAP 2 | 7% | 0.06 | 174 | 0.05 | 0 | |
| GAP 3 | 10% | 0.34 | 61 | 0.81 | 24 | |

Fig. 3.
Optical image of GAP-based network cured under the single curing system and dual curing system: (a) via maleimide, PBMI, (b) via dual curing, IPDI and BPS, (c) via dual curing, IPDI and PBMI, 1.5%, (d) via dual curing, IPDI and PBMI, 2.0%, (e) via dual curing, IPDI and PBMI, 2.3%, (f) via dual curing, IPDI and HBP, 2.0%, (g) via dual curing, IPDI and HBP, 4.0%, (h) via dual curing, IPDI and HBP, 3.0%, (i) via dual curing, IPDI and PBMI, 0.9%.
3.2 GAP-based networks cured under dual (hetero-) curing system
3.2.1 Curing via urethane and triazoline formation
To mitigate the mechanical limitations commonly associated with azide-alkene-only cured polymers -particularly the presence of free dangling chains-, we adopted an orthogonal (dual- or hetero-) curing strategy. The terminal hydroxyl groups of GAP were initially reacted with diisocyanate, IPDI, to form urethane bonds, providing linear connectivity as shown in Fig. 4, where a peak at 2260 cm-1 corresponding to the isocyanate group (-NCO) disappeared in 19 hours . Concurrently, the pendant azide groups were crosslinked through azide-alkene cycloaddition using difunctional maleimide compounds, MPBMI and PBMI, thereby extending the polymer network into a three-dimensional architecture.
Although maleimide compounds alone were earlier shown to be ineffective for network formation due to triazoline instability, under this hybrid system, they could be utilized in lower concentrations sufficient to form effective crosslinking structures without compromising thermal stability. As shown in Fig. 3(c), (d), (e), well-structured GAP-based polymer network was successfully achieved.
The mechanical properties of the resulting networks, presented in Table 2, indicate excellent performance. As expected, the minimum effective amount of maleimide required for curing varied depending on the chemical structure. For instance, MPBMI, containing two phenyl rings, achieved robust mechanical properties even at 0.9 wt%, indicative of highly efficient crosslinking; likewise, as illustrated in Fig. 3(b), the alkyne-based compound, BPS, demonstrated comparable performance at the same loading level, further confirming its effectiveness in network formation according to the data in Table 1. In contrast, PBMI, with only one phenyl ring, produced a soft and tacky material under identical loading, preventing accurate mechanical testing as shown in Fig. 3(i). Furthermore, as the maleimide content increased, the number of crosslinks between GAP side chains also increased, resulting in higher stress at max. strain but reduced strain at max. stress - consistent with the expected trade-off between rigidity and flexibility in polymer networks.
While this dual curing approach -especially involving maleimide derivatives for azide-alkene cycloaddition- successfully generated robust polymer networks, it could not completely eliminate gas bubble formation, as observed in Fig. 3(c), (d), (e). These bubbles are likely the result of thermal decomposition of the triazoline rings during prolonged curing at 50°C, underscoring the need for further optimization of alkene-based curing components.
Table 2.
Mechanical properties of dual (hetero-) cured GAP-based networks via side-linked triazolines (or triazoles).
3.2.2 Asymmetric curing via urethanes and triazole formation
To improve the structural stability and mechanical performance of GAP-based binders beyond the limitations observed in triazoline-cured system, we developed a novel curing strategy utilizing an asymmetric difunctional compound bearing both hydroxyl and alkyne functionalities, HBP. As illustrated in Scheme 4, the terminal hydroxyl group of GAP were first reacted with one isocyanate group of a diisocyanate molecule, IPDI, resulting in GAP chains terminated with free isocyanate groups. These terminal isocyanates subsequently reacted with the hydroxyl group of the asymmetric bifunctional compound to form urethane linkages, thereby incorporating the alkyne-functional end of the crosslinker into the network.
This design enables the pendant azide groups of GAP to undergo 1,3-dipolar cycloaddition with the alkyne moieties of the incorporated crosslinkers, effectively linking both the chain ends and side chains through a singly molecular bridge. While this curing method is not a classical orthogonal system involving two fully independent reactive pathways, it achieves selective, spatially resolved crosslinking that results in a highly integrated three-dimensional polymer network. As shown in Fig. 3(f), (g), and (h), the network architecture provided significantly enhanced structural coherence compared to binders cured via maleimide-based azide-alkene reactions. The observed improvement is attributed to the formation of thermally stable triazole rings in combination with robust urethane linkages.
Furthermore, as shown in Table 3, increasing the concentration of HBP crosslinker led to higher density, which is reflected in the typical elastomeric behavior: increasing stress at max. strain and modulus with decreasing strain at max. stress. These results are consistent with conventional polymer network theory, where increased crosslinking restricts chain mobility while reinforcing mechanical strength like stress at max. strain.
Table 3.
Mechanical properties of GAP-based networks via the selective dual-site crosslinking with an asymmetric difunctional compound.
3.2.3 Effect of dipolarophile structure on network properties
Due to difference in molecular weight and functionality, the actual weight of each dipolarophile required to achieve this index varied. For instance, the oligomeric dipolarophile DPCL required the highest mass addition, whereas low-molecular weight compound such as BPS and EDP required the least. Although BPS and EDP have comparable molecular weight, HBP bearing only one alkyne group per molecule also required a relatively larger amount to maintain the same equivalent index.
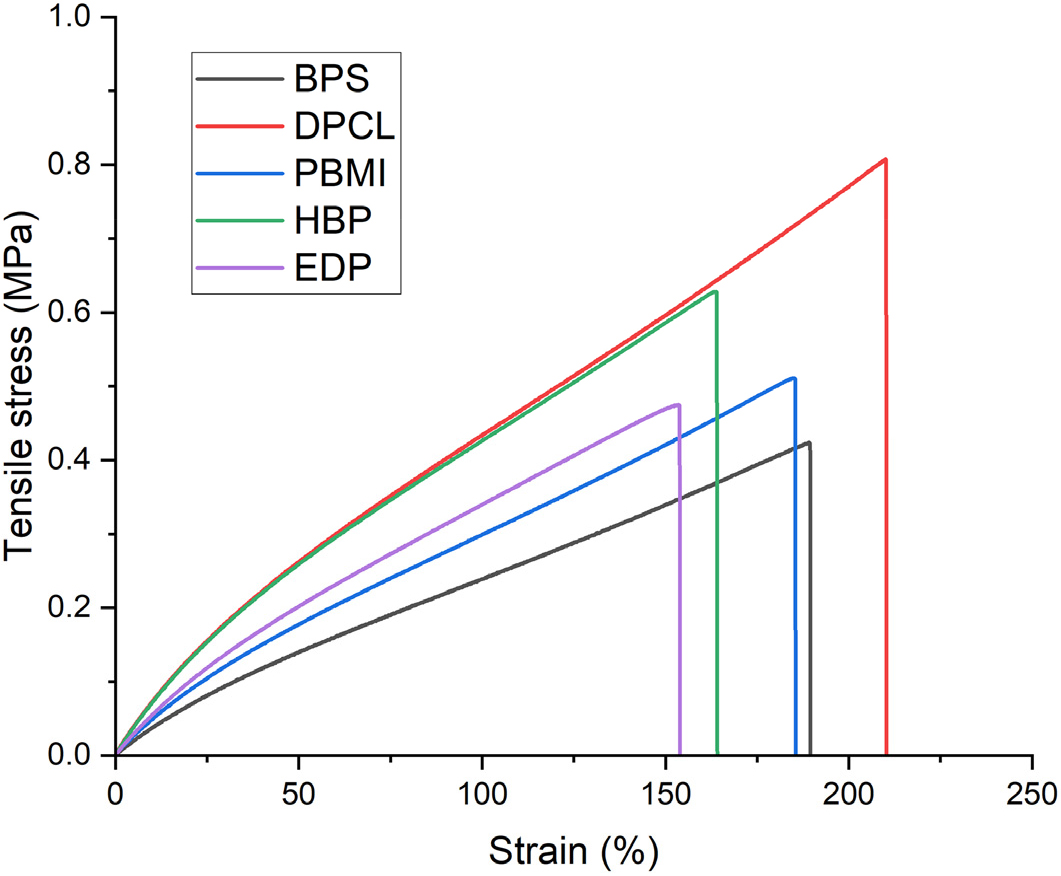
As shown in Fig. 5, the binder cured with DPCL exhibited the most superior mechanical performance among all dipolarophiles tested, likely due to its flexible oligomeric structure and enhance chain entanglement. Binders via BPS and EDP conveyed comparable mechanical performance, with BPS showing slightly better strain at max. stress. However, it is worth noting that the alkyne functionality in EDP is highly reactive toward azide groups, which can induce premature viscosity build-up during processing - posing a limitation for practical binder formulation. Considering both reactivity and mechanical balance, BPS appears to be the most effective dipolarophiles among those estimated.
Interestingly, the maleimide-based binder, despite involving a thermally less stable triazoline structure, exhibited unexpectedly strong mechanical properties, including both stress and strain. In addition, the HBP-based networks cured via an asymmetric curing route (i.e., one functional group reacting with GAP termini and the other with side-chain azides) showed notably high stress, further demonstrating the versatility of such difunctional crosslinking strategies.
Overall, the binder formed with DPCL was found to provide the best mechanical performance. However, both maleimide- and HBP-based networks also exhibited sufficient mechanical integrity to be considered viable energetic binder candidates for propellant formulations. It should be noted that although binders cured with BPS and EDP were mechanically less robust, their significantly lower loading levels imply minimal compromise in energetic content. This highlights a potentially valuable trade-off between mechanical performance and energetic efficiency. In support of this, the BPS-based with only 0.9 wt% dipolarophile content exhibited excellent elongation, as demonstrated in Table 2, confirming its practical potential.
4. Conclusion
GAP-based curing strategies were systematically explored by leveraging its dual reactive functionalities -terminal hydroxyl and pendant azide groups. Initial single curing system using difunctional alkynes or alkenes led to network formation via triazole or triazoline side-linkages; however, the resulting binders exhibited either insufficient mechanical properties or, in case of maleimide-based system, poor processability due to incomplete curing and gas evolution. To overcome these limitations, dual-site curing was implemented by combining urethane linkages at the polymer termini with azide-dipolarophile cycloadditions along the backbone, resulting in robust three-dimensional networks. An alternative asymmetric curing method utilizing a hydroxyl-alkyne bifunctional compound enabled efficient integration of chain-end and side-chain reactivity, yielding high-strength binders. Comparative analysis of dipolarophile structures-normalized by equivalent azide reactivity-revealed that while the oligomeric alkyne imparted excellent mechanical performance, their high required loading reduced energetic density. In contrast, the symmetric ester-functionalized alkyne demonstrated optimal trade-offs between mechanical integrity, reactivity and energetic contribution, making this the most practical dipolarophile candidates for advanced GAP-based energetic binder systems. This work contributes to the development of energetic GAP-based solid propellants for high-performance solid rocket motors, owing to the incorporation of highly energetic and thermally stable triazole moieties. Furthermore, the dual-crosslinked GAP-based networks show strong potentials as leading candidates for solid fuels in solid-ramjet or hybrid propulsion systems, thanks to their high density and elevated regression rate.