1. Introduction
2. Materials and Methods
2.1 Materials
2.2 Method for evaluating storage stability
3. Results and Discussions
3.1 Method Validation
3.2 Storage Stability Results
4. Conclusions
1. Introduction
A hypergolic propellant system involves a fuel and an oxidizer that ignite spontaneously upon contact, obviating the need for an external ignition mechanism. This unique property simplifies the propulsion architecture and reduces the risk of ignition failure, thereby improving overall system reliability. As a result, hypergolic propellants have been extensively used in spacecraft propulsion for decades. A widely adopted hypergolic combination is monomethyl hydrazine (MMH) as fuel and nitrogen tetroxide (NTO) as oxidizer, valued for its high ignition performance and long-proven reliability [1]. However, due to the severe toxicity and environmental hazards of this propellant combination, its use is now heavily regulated. Consequently, recent research has shifted toward alternative oxidizers with lower toxicity, such as high-concentration hydrogen peroxide and nitric acid-based oxidizers, including white fuming nitric acid (WFNA) and red fuming nitric acid (RFNA) [2,3,4,5,6].
Among these, hydrogen peroxide is a well-known liquid oxidizer that exhibits hypergolic behavior with certain fuels. Initially, it was used as a monopropellant to power the turbopumps of the V-2 rocket and has since been utilized in various propulsion systems. Furthermore, hydrogen peroxide functions as both a fuel and an oxidizer, enabling its use in both monopropellant and bipropellant systems. Nevertheless, the relatively high freezing point and susceptibility to decomposition of hydrogen peroxide can lead to significant issues during long-term storage. For example, 95 wt% hydrogen peroxide, a typical concentration used in propulsion, has a freezing point of approximately -5.6°C, which is considered high under typical operating conditions. Such characteristics can pose risks in space environments, where freezing may lead to ignition failure or even catastrophic malfunction.
To address these challenges, several studies have sought to improve the performance of hydrogen peroxide through the incorporation of additives. Weiser [7,8] proposed an oxidizer formulation in which ammonium dinitramide (ADN) and ammonium nitrate (NH4NO3) were dissolved in high-concentration hydrogen peroxide to develop a greener alternative to hydrazine-based hypergolic propellants. Ignition performance was assessed using 1-allyl-3-methylimidazolium dicyanamide (AMIM DCA) as the fuel. The addition of ADN and NH4NO3 was found to lower the freezing point of the resulting solution. Although these additives contributed to freezing point depression, their incorporation resulted in a slight increase in ignition delay time (IDT) relative to pure hydrogen peroxide. Park [9] investigated hypergolic propellant systems in which nitrate-based additives (LiNO3 and NH4NO3) were dissolved in hydrogen peroxide to formulate modified oxidizers. Ignition performance was evaluated using ionic liquid fuels, EMIM SCN and BMIM SCN, and their physical properties were also investigated. The addition of nitrate salts significantly lowered the freezing point of 95 wt% hydrogen peroxide, and this effect was more pronounced with LiNO3 than with NH4NO3. In addition, compared to pure hydrogen peroxide, the ignition delay time (IDT) was considerably reduced as the concentration of LiNO3 increased. This result indicates the effectiveness of LiNO3 in enhancing ignition performance.
Most previous studies have focused on enhancing the propulsive and physical properties of propellant formulations. However, hydrogen peroxide is inherently unstable and readily decomposes, even in the presence of trace impurities [10]. This decomposition limits storage stability and reliability, potentially compromising mission performance in space propulsion applications. Therefore, assessing the compatibility between hydrogen peroxide and performance-enhancing additives is essential. This study aims to establish a quantitative method for evaluating high-concentration hydrogen peroxide formulations containing such additives and to investigate their long-term storage stability.
In this study, the effect of LiNO3 addition on the storage stability and ignition performance of hydrogen peroxide was examined. The hydrogen peroxide concentration in the prepared formulations was measured using a redox titration method. All samples were stored at 2°C for 100 days under refrigerated and controlled conditions. After storage, visual inspection, density measurement, concentration analysis, and ignition performance evaluation based on IDT measurements were conducted.
2. Materials and Methods
2.1 Materials
Hydrogen peroxide with a nominal concentration of 90 wt% was used in this study. The actual concentration, measured using a refractometer (PR-50HO, ATAGO, Japan), was determined to be 89 wt%. Lithium nitrate (LiNO3) was selected as an oxidizer additive based on its superior performance in a previous study [9]. It was purchased in high purity (99.9%) from Sigma-Aldrich (USA). LiNO3 was mixed with 90 wt% hydrogen peroxide to prepare solutions containing 1, 5, 10, and 20 wt% additive concentrations (Table 1). The mixtures were stirred at room temperature (25°C) for 1 hour, stirred homogeneous solutions without any undissolved residues. The fuels used for ignition performance evaluation after storage were EMIM SCN and BMIM SCN, identical to those in previous studies [9]. Both were used with a purity greater than 95%, as provided by Sigma-Aldrich.
Table 1.
Compositions of H2O2-based solutions with and without LiNO3.
| Formulation | H2O2 (wt%) | H2O (wt%) | LiNO3 (wt%) |
| H2O2 89 wt% | 89 | 11 | 0 |
| Li1 | 88.11 | 10.9 | 1 |
| Li5 | 84.55 | 10.5 | 5 |
| Li10 | 80.1 | 9.9 | 10 |
| Li20 | 71.2 | 8.8 | 20 |
2.2 Method for evaluating storage stability
2.2.1 Sample storage conditions
The samples were stored in 40 mL borosilicate glass vials under refrigerated storage conditions (2°C) for 100 days. The vials were sealed with polypropylene caps and polyethylene terephthalate (PET) liners. According to the literature, both the vial body and cap materials have been shown to be compatible with hydrogen peroxide [10]. Although compatibility data for the liner material are limited, no visible chemical reaction was observed during storage. The surface to volume (S/V) ratio is defined as the internal surface area of the container divided by the volume of the contained liquid. This value is known to inversely correlate with storage stability [11]. In the present investigation, an S/V ratio of 4.572 in-1 was employed, indicating a relatively severe storage environment in comparison with that of conventional hydrogen peroxide tanks, which generally exhibit a much lower S/V ratio (S/V ≈ 0.33 in-1[11]).
2.2.2 Concentration Measurement
The concentration of hydrogen peroxide is a key indicator for evaluating long-term storage stability. Common quantitative analysis methods include potassium permanganate (KMnO4) titration, refractive index measurement, and density measurement. Among these, refractive index and density methods allow rapid estimation due to their well-established linear correlation with peroxide concentration under standard conditions. However, the presence of significant amounts of additives can disrupt this linearity, leading to potential deviations in measurement accuracy. In contrast, KMnO4 titration determines the concentration based on the stoichiometric redox reaction between hydrogen peroxide and potassium permanganate, and is therefore less affected by the presence of interfering additives. Accordingly, KMnO4 titration was adopted in this study to quantitatively evaluate hydrogen peroxide concentration. The method is based on the stoichiometric redox reaction between KMnO4 and H2O2 in acidic media, as represented by the equation below.
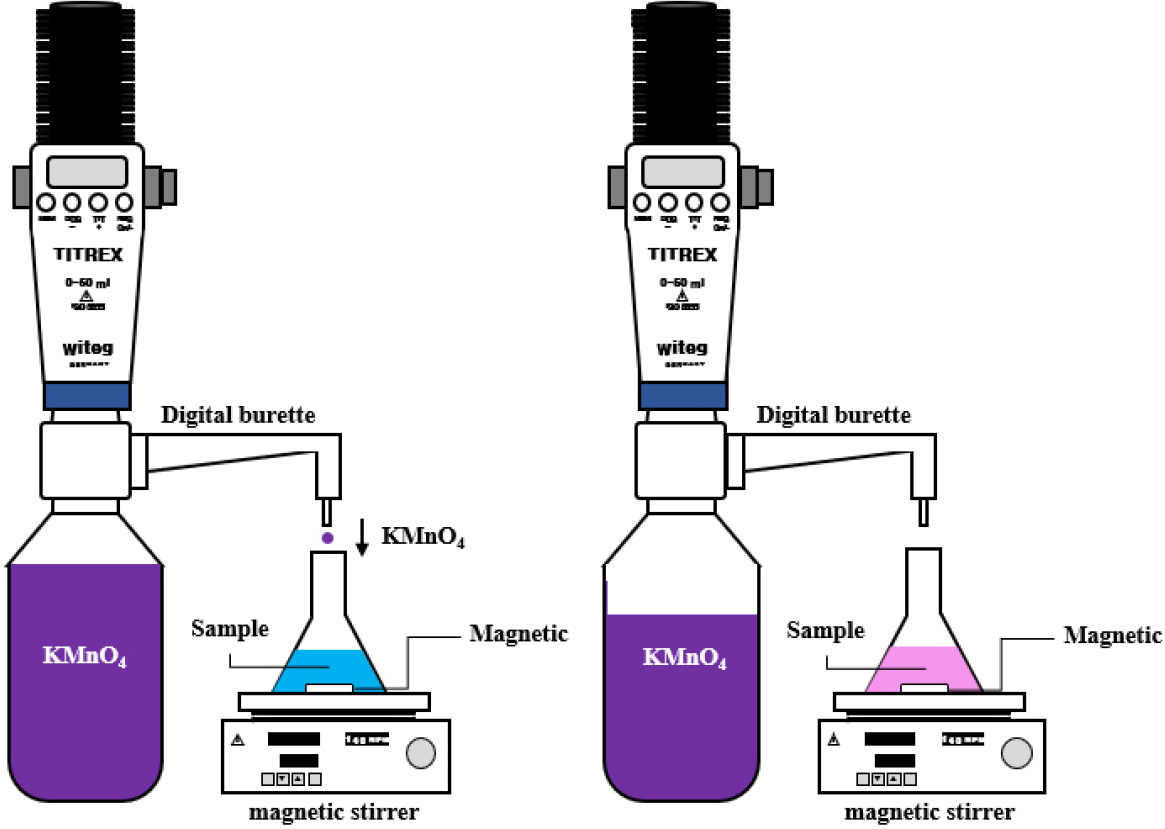
In this study, the hydrogen peroxide concentration was measured both before and after storage using KMnO4 titration to evaluate the degree of concentration loss. The titration procedure followed the protocol reported in previous studies [12]. Sulfuric acid (Samchun Pure Chemical Co., Ltd., Korea) was used to maintain the acidic environment during titration. An Eppendorf Multipette E3 was used for reagent preparation, and a standardized KMnO4 solution (0.02 mol/L, Samchun Pure Chemical Co., Ltd., Korea) served as the titrant. The experimental setup is shown in Fig. 1. A digital burette (WI.5.497.150, witeg Labortechnik GmbH., Germany) was used for precise titration. Samples were stirred using a magnetic stirrer and stir bar to ensure homogeneous mixing at ambient temperature (25 ± 1°C). Each measurement was performed four times per sample to improve data reliability.
2.2.3 Ignition Delay Time Measurement
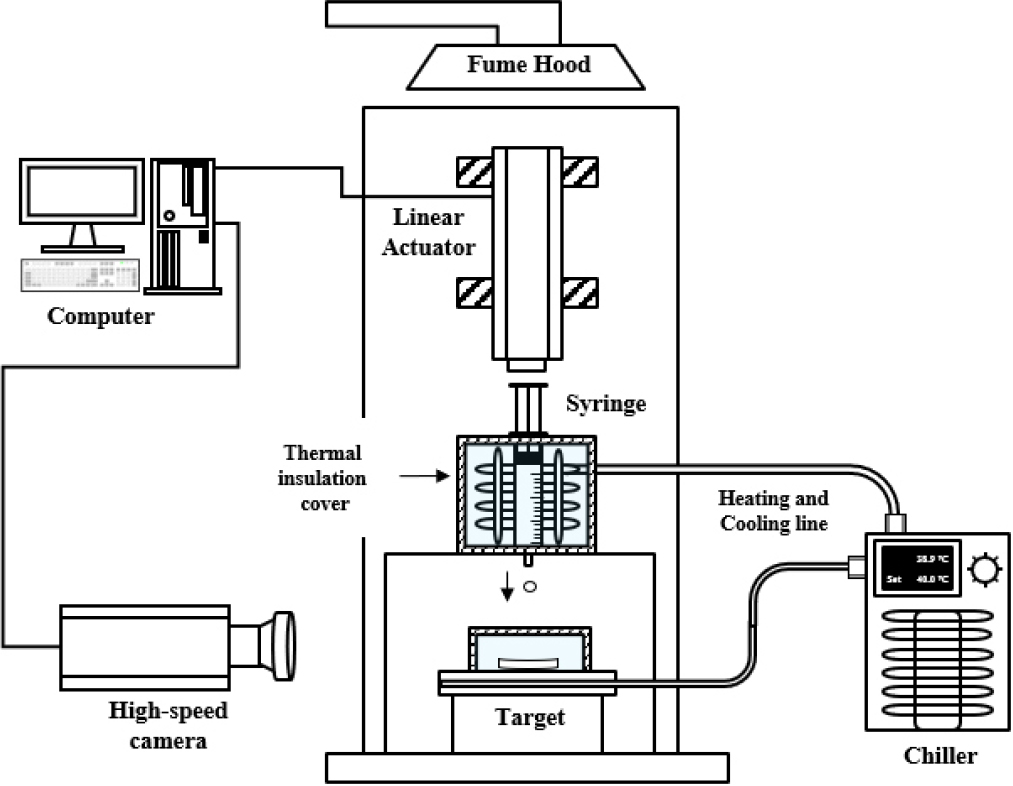
Ignition delay time (IDT) is a key metric for evaluating the ignition performance of hypergolic propellants. In this study, IDTs of samples stored for 100 days were compared with freshly prepared samples under identical initial conditions. Fresh samples were prepared by adjusting the initial concentration based on refractive index measurements. IDT was measured using a high-speed camera (VEO-E310L, Phantom, USA) operating at 2000 fps. It was defined as the time interval between the initial contact of the oxidizer droplet with the fuel and the onset of visible ignition. A schematic of the IDT measurement system is shown in Fig. 2. In this setup, oxidizer droplets were released by a linear actuator (Coolmuscle, Myostat Motion Control Inc., Canada) mounted at the top of the apparatus. Heat exchangers connected to the oxidizer and fuel reservoirs maintained their temperatures at 25°C prior to droplet release. The distance between the oxidizer and fuel reservoirs was fixed at 220 mm. Experiments were conducted under fuel-rich conditions with an oxidizer-to-fuel (O/F) mass ratio of 0.5. All experimental parameters were held constant, and each test was repeated four times to ensure data reliability.
3. Results and Discussions
3.1 Method Validation
3.1.1 Accuracy and Precision
In this study, the redox titration method was employed to determine hydrogen peroxide concentration in solutions with and without additives. However, if an additive directly participates in the redox reaction, titration results may not accurately reflect the actual H2O2 content. Therefore, a comparative validation was conducted to assess the method’s reliability.
For samples without additives, titration results were compared with values obtained using a refractometer (PR-50HO, ATAGO, Japan; accuracy ±0.5). Two H2O2 samples (90 wt% and 95 wt%) were analyzed. The refractometric values were 89.0 wt% and 94.95 wt%, while titration yielded 91.16 wt% and 97.31 wt%, resulting in relative errors of 2.43% and 2.49% (Table 2). In both cases, titration produced slightly higher H2O2 concentrations than refractometry, with comparable standard deviations for both methods.
Table 2.
H2O2 concentration comparison by titration and Refractometer ([-]: standard deviation).
| Formulation | refractometer (wt%) | Titration (wt%) | Error (%) |
| H2O2 89 wt% |
89 [0.59] |
91.16 [0.35] | 2.43 |
| H2O2 95 wt% |
94.95 [0.29] |
97.31 [0.46] | 2.49 |
For additive-containing samples, titration results were compared to theoretical values, as refractometry was not applicable due to optical interference. Samples containing 1, 5, 10, and 20 wt% LiNO3 in 89 wt% H2O2 were analyzed. Relative errors ranged from 1.73% to 2.49%, with a standard deviation of approximately 0.5 (Table 3). Overall, titration slightly overestimated H2O2 concentration relative to theoretical values, consistent with the trend in pure H2O2 samples. These results suggest that KMnO4 titration is suitable for evaluating H2O2 concentration even in the presence of LiNO3.
Table 3.
H2O2 concentration comparison by titration and theoretical value ([-]: standard deviation).
| Formulation | Theoretical value (wt%) | Titration (wt%) | Error (%) |
| L1 | 88.11 |
90.21 [0.69] | 2.39 |
| L5 | 84.55 |
86.01 [0.12] | 1.73 |
| L10 | 80.1 |
81.52 [0.2] | 1.78 |
| L20 | 71.2 |
72.94 [0.14] | 2.45 |
This overestimation may occur because the concentration is determined based on the titration endpoint rather than the equivalence point. The equivalence point refers to the exact stoichiometric point at which the moles of titrant and analyte are equal, whereas the endpoint is the observable signal (e.g., color change) indicating the completion of the titration. In redox titrations using KMnO4, the endpoint typically appears slightly after the equivalence point, potentially resulting in excess titrant addition and a slight overestimation of the analyte concentration.
3.1.2 Reproducibility
Because storage stability testing requires long-term monitoring, ensuring reproducibility is essential to validate the reliability of the analytical method. To evaluate reproducibility, the titration measurements described in Section 3.1.1 were repeated over a three-day period. The standard deviation of the relative errors between the titration results and the reference values (from refractometry or theoretical estimates) ranged from ±0.02 to ±0.33, regardless of the presence or amount of LiNO3. These results indicate that the redox titration method yields consistent measurements even in the presence of additives, enabling reliable tracking of H2O2 concentration during extended storage.
3.2 Storage Stability Results
3.2.1 Visual Inspection
Fig. 3 shows the visual appearance of each oxidizer sample 100 days before and after storage. The top images depict the samples prior to storage, while the bottom images correspond to those stored at 2°C for 100 days. No visible changes such as precipitation or turbidity were observed in any of the samples.
3.2.2 Concentration and Density
The density and concentration of each oxidizer sample before and after storage are summarized in Table 4. After 100 days of storage, all samples showed a decrease in hydrogen peroxide concentration, ranging from 0.46 to 1.07 wt%, due to gradual decomposition over time. Similarly, the density of all samples also decreased, with changes ranging from 0.002 to 0.013 g/cm³. The extent of concentration and density loss was comparable across all samples, regardless of the presence or amount of LiNO3. This suggests that the addition of LiNO3 does not negatively affect the storage stability of hydrogen peroxide. Additionally, it should be noted that the addition of LiNO3 reduces the absolute H2O2 content in the solution.
In general, higher concentrations of hydrogen peroxide are known to exhibit superior storage stability, whereas lower concentrations tend to decompose more readily, leading to reduced stability [11]. Therefore, the comparable concentration loss across all samples further supports that LiNO3 does not promote degradation. Interestingly, the sample containing 10 wt% LiNO3 exhibited the smallest losses in both concentration and density. The absolute concentration loss in this sample was less than half of that observed in the pure hydrogen peroxide sample. This behavior may be attributed to the physicochemical effects of LiNO3 on the H2O2-H2O system. At an intermediate concentration of approximately 10 wt% LiNO3, lithium and nitrate ions may stabilize the hydrogen-bonding network of the solvent, thereby reducing the molecular mobility of H2O2 and suppressing its self-decomposition pathways. Consequently, the mitigated decreases in concentration and density observed for the L10 formulation are likely derived from an optimal balance between H2O2 stability and additive concentration. However, further investigation is required to quantitatively confirm this effect.
Table 4.
Changes in H2O2 concentration and density of each formulation 0 day and 100-day storage ([-]: standard deviation).
3.2.3 Ignition Delay Time
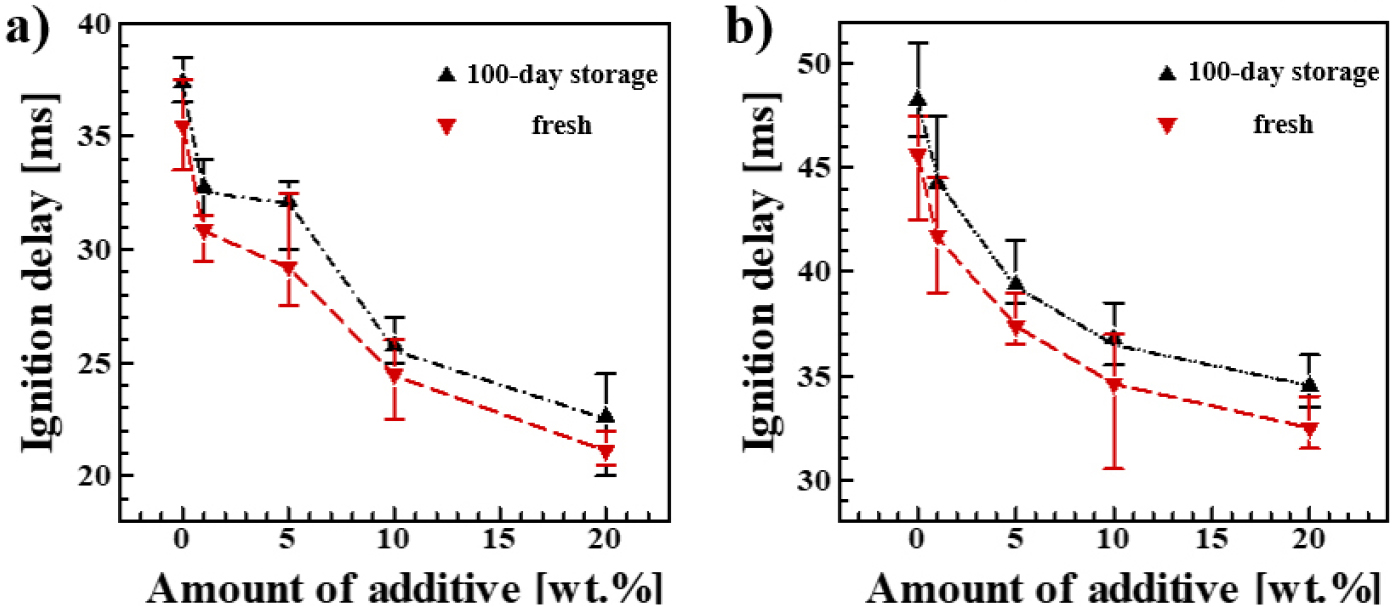
Fig. 4 shows the IDT measurements for each propellant combination after 100 days of storage, along with those of the freshly prepared (Fresh) samples. All stored samples showed an increase in IDT. This trend was consistently observed across all fuel groups, including both EMIM SCN and BMIM SCN. The increase in IDT is attributed to the reduction in hydrogen peroxide concentration during storage. This concentration decrease was also confirmed by the titration results. The trend of decreasing IDT with increasing LiNO3 content was maintained even after storage. Moreover, the extent of IDT increase was similar across all additive levels, indicating that LiNO3 does not adversely affect ignition performance during storage.
4. Conclusions
In this study, the storage stability of hydrogen peroxide containing lithium nitrate (LiNO3) was evaluated through quantitative measurements of ignition delay time, density, and concentration, along with qualitative visual observations. The KMnO4-based titration method exhibited sufficient precision and reproducibility in measuring the concentration of hydrogen peroxide containing lithium nitrate, demonstrating its suitability for storage stability assessment. Each sample was stored at 2°C for 100 days. After storage, both the additive-containing and pure hydrogen peroxide samples exhibited similar reductions in concentration and density. Among them, the L10 formulation (H2O2 + 10 wt% LiNO3) exhibited the least degradation. The decrease in hydrogen peroxide concentration was less than half that observed in the pure sample. This result suggests improved storage stability. However, due to potential measurement uncertainties, extended long-term evaluation is required to confirm this effect. IDT measurements revealed increased ignition delays in all stored samples, likely due to reduced peroxide concentration. Overall, the presence of LiNO3 did not adversely affect either storage stability or ignition performance. These results indicate that LiNO3 is a promising additive for enhancing the performance of hydrogen peroxide without compromising its storability. It should be noted, however, that this study was conducted under a single temperature condition of 2°C and a relatively short storage period of 100 days. To more clearly elucidate the effects of the additive, additional evaluations over a wider temperature range are required. Future work will focus on long-term storage tests to validate the reliability of the present findings.