1. Introduction
2. Experimental Setup
2.1 Synthesis and processing of LaCoO3/rGO nanocomposites
2.2 Material characterization
2.3 Shock wave loading technique
3. Results and Discussion
3.1 X-ray diffraction analysis
3.2 FESEM analysis
3.3 Raman spectra analysis
3.4 FTIR spectra analysis
3.5 PL spectroscopy analysis
3.6 Isothermal magnetization analysis
4. Conclusions
1. Introduction
Nanotechnology has garnered significant attention for its broad range of applications, including photocatalytic, energy, environmental, disease diagnostics, therapeutic purposes, and storage applications. When compared to the bulk materials, the nanomaterials scale down to nanoscale and exhibit the salient features in optical, magnetic, mechanical, and electrical behaviours [1,2]. Nanoparticles that are composed of transition metal oxides have been extensively investigated and implemented in various disciplines. Particularly, perovskites are the metal oxide materials that have been represented in the formula ABO3, because these materials are important due to their attraction in photochemical, ferroelectric, optical, and conduction properties [3,4]. Among the perovskite materials, lanthanum cobalt oxide LaCoO3 (LCO) is one of the most important, having been used for its catalytic activity, thermal stability, good oxidation behaviour, and superconducting properties. Interestingly, LaCoO3 exhibits diamagnetic behavior at low temperatures, but above around 100°C it shows paramagnetic behavior due to a spin-state transition of Co3+ ions [5]. Moreover, the magnetic properties of LCO have been reported widely, and the investigations have demonstrated that the effective magnetic moment is significantly lower than the spin values for the higher spin of the Co3+ ion [6]. Further, the materials are used in spintronic device fabrication due to their higher Curie temperature (TC) and higher symmetry structure [7]. Due to its insulating and ferromagnetic nature, the material is a viable choice for making the non-dissipative spintronic devices [8,9].
The magnetic and electrical properties of LaCoO3 (LCO) change significantly when it is doped/composited with alkaline-earth or transition metal elements. Pure LCO shows diamagnetic behaviour at low temperatures but becomes paramagnetic above about 35 K. When doped with Sr, Ba, or Ca, the material can develop ferromagnetic properties at certain compositions [10,11,12,13,14,15,16,17]. This ferromagnetism is linked to the intermediate-spin (IS) state of Co3+ ions and structural distortions in the crystal lattice. Such distortions play an important role in LCO’s unusual magnetic behaviour, although its exact crystal structure is still under debate [17,18]. Ferromagnetic ordering has been observed around 85 K in differently synthesized LCO samples, while some studies report a non-magnetic state. The low-temperature ferromagnetism is believed to result from short-range interactions between Co3+ and Co4+ ions, super exchange interactions at the surface, and the presence of a small number of high-spin states [19,20,21,22]. Sanchayita Nag et.al reported the enhanced coercivity value in the room temperature as well in low temperature for Ni0.5Zn0.5Fe2O4/rGO [23]. This suggests that the adding of rGO influences the magnetic property. When used as a support material in composite formation, reduced graphene oxide (rGO) enhances the stability, adsorption capability, and electronic conductivity [24,25]. Hence the combination of LCO/rGO may results in introduction of local lattice distortions and oxygen vacancies. The results in the composite helps to stabilization of high spin Co state, reason to ferromagnetic nature at room temperature. Adding rGO as a composite, not only improves the structural, electronic structure of LaCoO3, but also functions of magnetic coupling of composites resulting in higher saturation magnetization, coercivity and overall magnetic performance.
In recent decade, our group focused on the impact of shock waves on bulk and nanocrystalline materials and explored some interesting results. In particular, researchers have widely studied nanocrystalline materials, and previous reports have documented this [26,27]. Among the reported results, under the shocked conditions, the ferrite material of CoFe2O4 has shown the hard-to-soft magnetic properties [28]. Meanwhile, there are no changes in electronic and crystallographic magnetic phase transitions that occurred for NiFe2O4 by the impact of shock waves. NiFe2O4 ferrites confirm that the material is highly stable to the shock wave [29]. For AgO nanoparticles under shocked conditions, the particle size increases from 72 nm to 162 nm as the number of shocks increases from 0 to 200, confirming that the value of saturation magnetization is reduced from 2.183 emu/g (0 shocks) to 1.416 emu/g (200 shocks) [30]. Further, Fe2O3 nanoparticles were systematically studied from 0 to 150 shock pulses; the material’s magnetic behaviour was stable up to 100 shocks, and it lost stability at 200 shock pulses. Here, the ferromagnetism of Fe2O3 is systematically reduced due to applying shocks [31]. MnO2 nanorods were synthesized using the hydrothermal technique and investigated under shock conditions. The results clearly mention that no changes in crystallographic phase transition and not even in grain boundaries have been observed. But, the noticeable changes recorded in saturation magnetization and coercivity due to spin canting are due to sudden releases of pressure and temperature induced by the impact of the shock wave [32].
Based on the literature, the purpose of the research is to investigate the magnetic and structural modification of LaCoO3/rGO nanocomposites. Because there is no comprehensive analysis of perovskite-based composites reported. Applying shocks to the materials induces the lattice distortions, creates the oxygen vacancies, and develops the interfacial interaction between the LaCoO3/rGO nanocomposites. Hence, we systematically investigated the impact of shock waves on LaCoO3/rGO for understanding the crystallographic and magnetic phase transition.
2. Experimental Setup
2.1 Synthesis and processing of LaCoO3/rGO nanocomposites
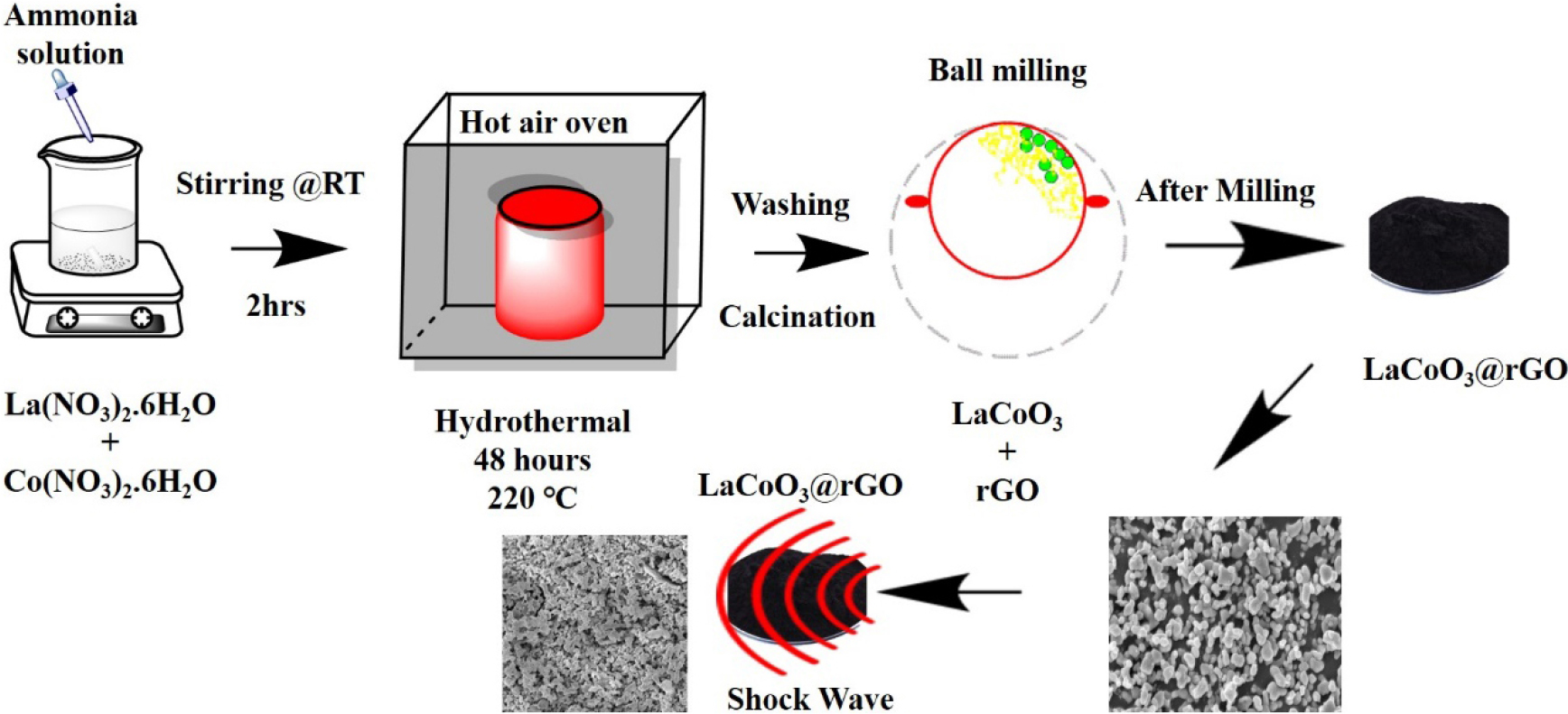
For the preparation of LaCoO3/rGO nanocomposites, La (NO3)3・6H2O, (Co(NO3)2・6H2O), ammonia, and rGO were purchased without purification, and deionized (DI) water was used for the synthesis process. Lanthanum nitrate hexahydrate and cobalt nitrate hexahydrate were dissolved in deionized water and mixed thoroughly by magnetic stirring at room temperature (RT) for the perfect mixing. Further, ammonia is added to the mixer dropwise to maintain the basic pH values for about 2 hours for the homogeneity. The mixture was transferred to a Teflon cell for hydrothermal treatment for 48 hrs at 220°C. The materials cool down to RT, are washed with water and ethanol, and are then dried and calcined. Furthermore, the calcined material of LaCoO3 mixed with rGO under ball milling, and the final product is obtained as LaCoO3/rGO nanocomposites. The schematic diagram of the LaCoO3/rGO nanocomposites is shown in Fig. 1.
2.2 Material characterization
X-ray diffraction (XRD) analysis was performed in the range of 10-80° using a multi-purpose X-ray diffractometer (XRD-6000, Shimadzu, Kyoto, Japan) at the Intelligent Construction System Core-Support Center, Keimyung University. To analyze the morphology of the control and shocked samples, the ZEISS sigma (Carl Zeiss AG) model is used. The FTIR spectra were recorded using a Perkin Elmer spectrometer (PerkinElmer, USA) in the range of 4000-400 cm-1, and the Horiba Fluorolog-3 spectrofluorometer was used to obtain a PL spectrum (Horiba Scientific, Japan) with the excitation wavelength at 325 nm. Raman spectra is used to analyze the vibrations and phonon modes, using the Renishaw inVia Raman microscope (Renishaw, UK) equipped with a 532 nm laser as the excitation source. The isothermal magnetization measured through VSM-PPMS (model 6000, USA) to analysis the magnetic properties of control and shocked samples.
2.3 Shock wave loading technique
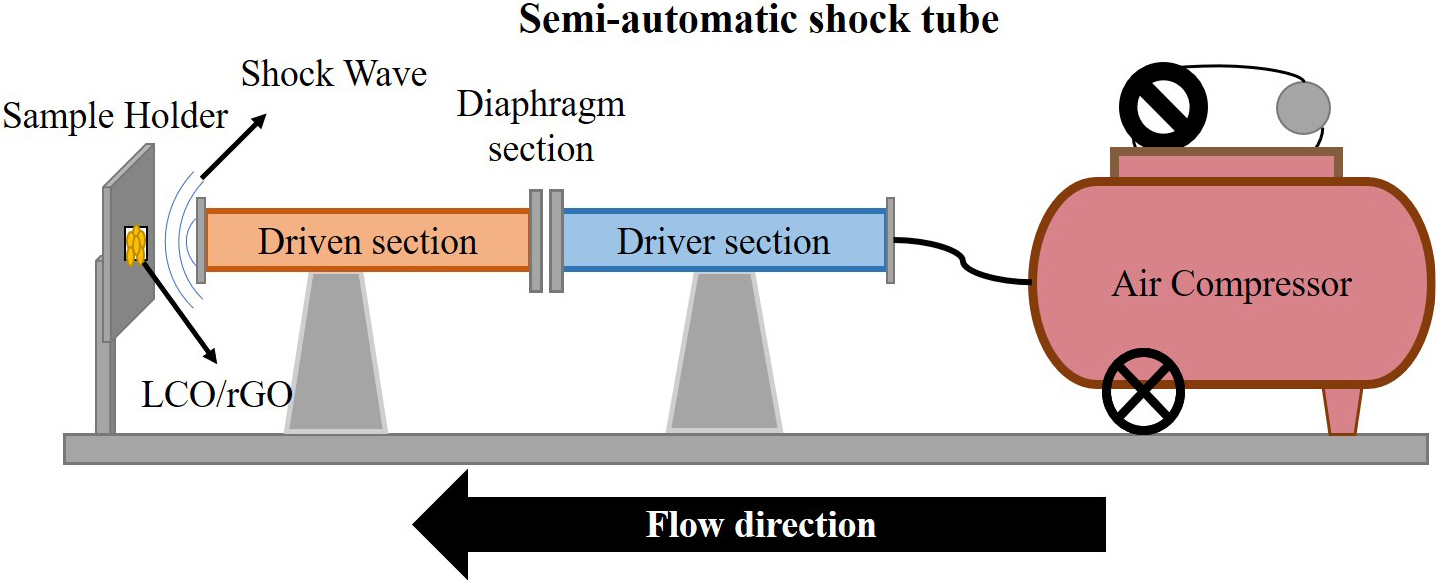
The semi-automatic, manually operating Reddy shock tube was used to apply the shock pulses to the LaCoO3/rGO nanocomposites. Like a conventional shock tube, the semi-automatic tube consists of three parts: the driver section (high-pressure gas), the driven section (low-pressure gas), and the diaphragm section (100 gsm mylar sheet). The pressure in the driver section is 8 bar, and the driven section is kept at atmospheric pressure. For the shock wave study, the sample of LaCoO3/rGO was divided into three for control, 100, and 200 shock pulses. The schematic diagram of the shock tube is mentioned in Fig. 2, and the dimensions of the tube are 1.5 cm as an inner diameter and 2 cm as an outer diameter. As mentioned in the schematic diagram, the sample is positioned 1 cm away from the driven tube to apply continuous shock pulses. The diaphragm, 100 GSM paper, ruptures when the driver section’s pressure reaches a critical pressure (8 bar) and shock wave as a pulse is generated and propagates through the driven section. The sample is positioned in a custom-made sample holder which is placed 1 cm away from the open end of the driven section, ensuring that the shock pulse impacts the sample without significant energy loss due to divergence.
3. Results and Discussion
3.1 X-ray diffraction analysis
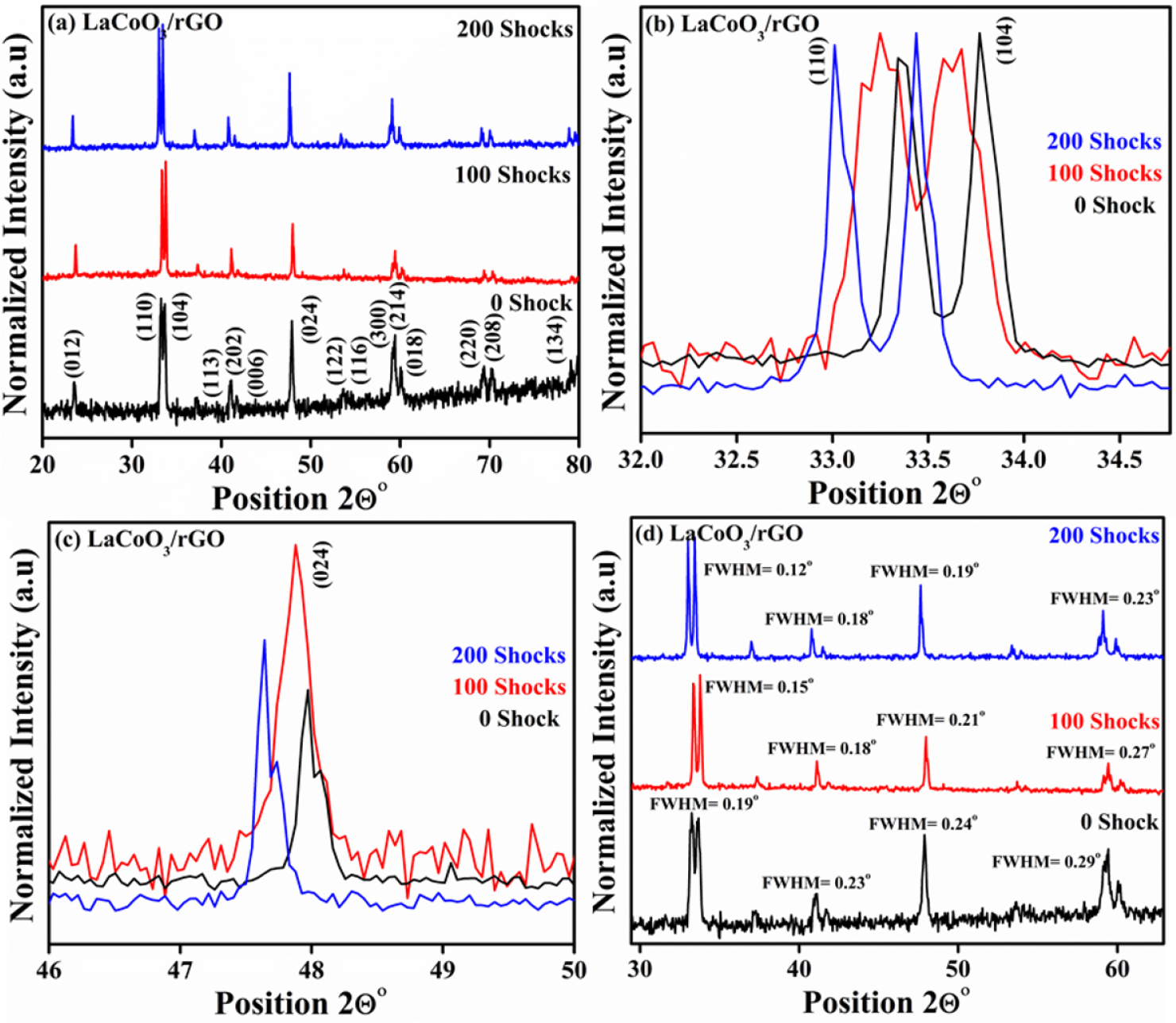
The powder X-ray diffraction of the LaCoO3/rGO nanocomposite is shown in Fig. 3, and the crystal structure of the perovskite is generally predicted using synthesis techniques. The materials are subjected to different shock pulses, such as 100 and 200 shocks, to understand crystallographic stability. Here, the XRD pattern reveals the hexagonal crystal structure with space group of up R -3 c H (167), which is highly matched with the JCPDS card No. 48-0123. The observed peaks of control samples, such as (012), (110), (104), (113), (202), (006), (024), (122), (116), (300), (214), (018), (220), (208), and (134), confirm the material is LaCoO3 perovskite and has a good crystalline nature [33].
Furthermore, the XRD peaks of rGO are not visible in the pattern due to the higher crystallinity of LaCoO3 perovskite, and no extra peaks are observed, confirms the materials are stable with the applied shock pulses.
The peaks shifting to lower angles under shocked conditions confirm the lattice distortions in the crystal system due to a sudden transient temperature and pressure from shock waves. Further, a small change in the intensity leads to the dynamic recrystallization of the LCO/rGO nanocomposite.
Additionally, the peaks of (110), (104), and (024) are considered to understand the shifts; the results mention that all peaks shift slightly towards lower angles, confirming lattice expansion is shown in Fig. 3(b) and (c)[34,35].
Furthermore, FWHM (full width at half maximum) was calculated for control and shocked samples as shown in Fig. 3(d). Interestingly, the FWHM slightly decreases as the number of shock pulses increases from 0 to 200 shocks. For the plane of (110) and (114), the FWHM value is 0.19o for 0 shock and decreased to 0.12o for 200 shocks. Similarly, the FWHM is decreased from 0.24o to 0.19o for the (202) and 0.29o to 0.23o for the (214) planes. The decreases in FWHM under shocked conditions induce the dynamic recrystallisation and enhance the crystalline nature. The lattice parameter and crystalline size are calculated for the control and shocked samples, and the values slightly increase with increasing the shock pulses from 0 to 200, and the calculated values are mentioned in Table 1. The values of the lattice parameter and crystalline size increased due to the shift of the XRD peaks towards a lower angle and decreased the FWHM under shocked conditions.
Table 1.
Lattice parameter and crystalline size of control and shocked samples of LCO/rGO nanocomposites.
3.2 FESEM analysis
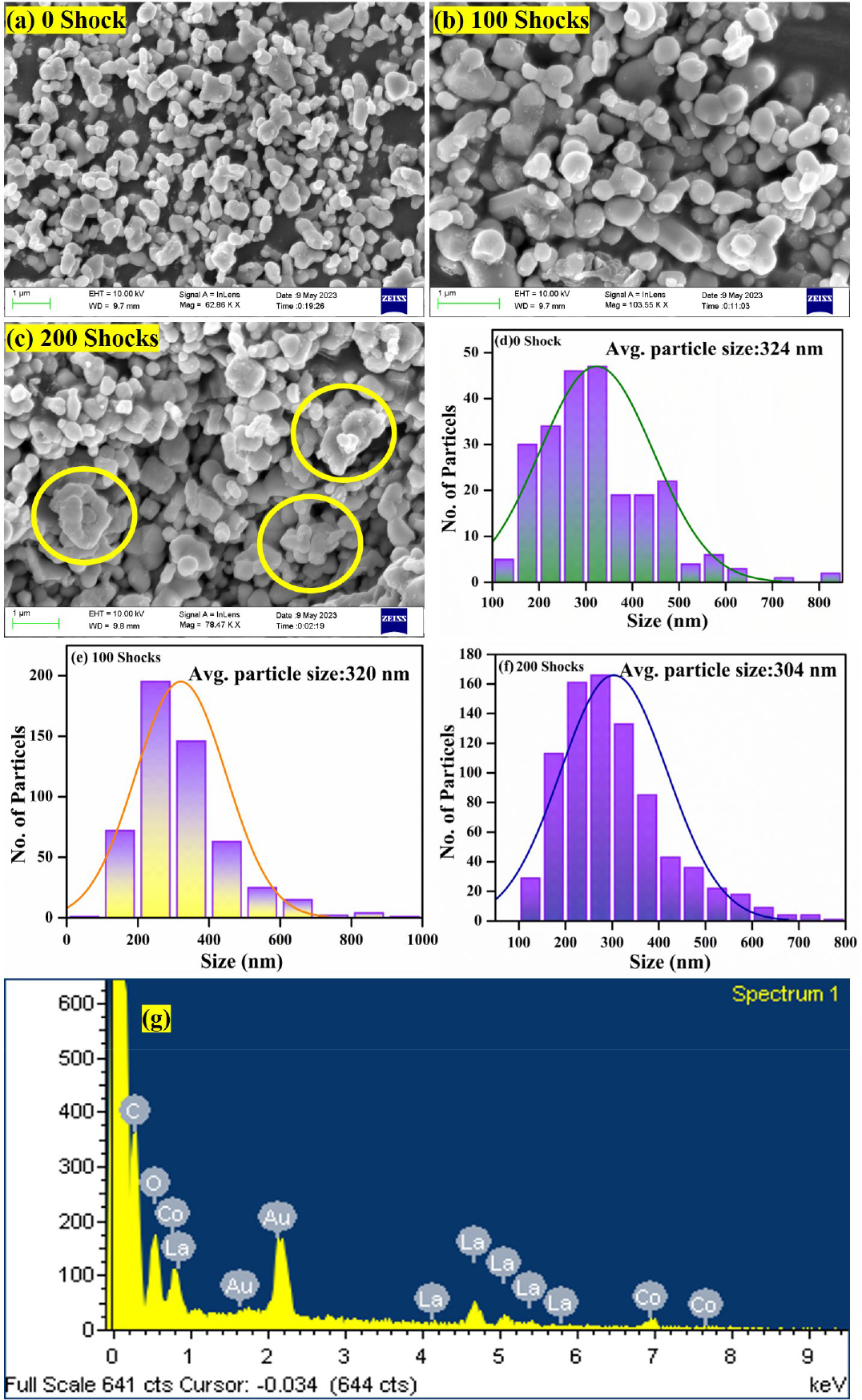
Fig. 4(a)-(e) shows FESEM with histogram analysis of LCO/rGO nanocomposites for control and shocked samples. At 0 shock, the morphology seems like spherical particles and the average of the particle is 324 nm. Due to smaller amount of rGO in LCO, the sheets are not clearly visible in the morphology. At 200 shocks, the sample retain their original structure and the size of the particles is slightly reduced to 304 nm due to shock wave. When the shocks propagate thorough the materials, shocks induce the rapid heating in temperature, it enhances the pressure leads to disrupting the crystal lattice and internal stress. Interestingly at 200 shocks, the materials surface is melted slightly and mentioned in the yellow circle. Because the sudden releases of energy in terms of pressure and temperature melting the materials and attributed to the dynamic recrystallization. Even though, the findings confirm the materials stability and the robustness structure of the LCO/rGO nanocomposites. EDAX analysis is shown in Fig. 4(f), which confirms the presence of La, Co, O and C elements in the composites.
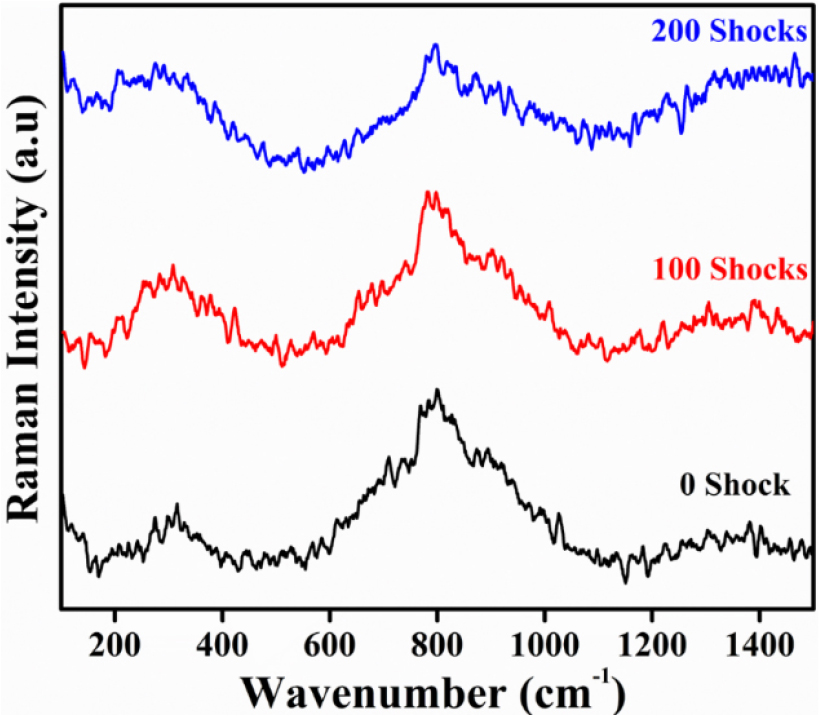
3.3 Raman spectra analysis
The Raman spectra of LaCoO3-rGO composites show characteristic phonon modes of LaCoO3 in the 100-900 cm-1 region, corresponding to La-O vibrations, Co-O bending, and Co-O stretching is shown in in Fig. 5 for control and shocked samples. The prominent peak for LaCoO3 is also observed by Jhelai et al. [16]. In the LA-RGO sample, these bands are distinct and relatively sharp, confirming the preservation of crystallinity and a stable lattice with minimal background interference. The LA-100 sample also shows clear phonon features, though with slightly broader peaks, indicating that shock wave introduces some disorder but does not significantly damage the lattice. In contrast, the LA-200 spectrum exhibits broadened peaks with an elevated baseline, which is consistent with strong disorder, defect formation, and possible oxygen vacancies. This suggests that excessive shock pulses disrupt the perovskite structure and increase the electronic background or fluorescence. The D band around 1350 cm-1 is visible; however, the G bands of rGO are not seen in the 1300-1600 cm-1 area. This could indicate either a poor rGO contribution in comparison to LaCoO3. Sivakumar et al., reported the reduction of carbon clusters in Raman spectroscopy with the increase of shock wave [36]. Overall, the data show that when LaCoO3 is coupled with rGO and exposed to low shock (100), it retains its phonon structure, but larger shock (300) causes significant defect states and lattice disorder. This suggests that while high shock wave damages crystallinity and may adversely influence the composites functional qualities, moderate shock maintains the structural integrity of the material [37].
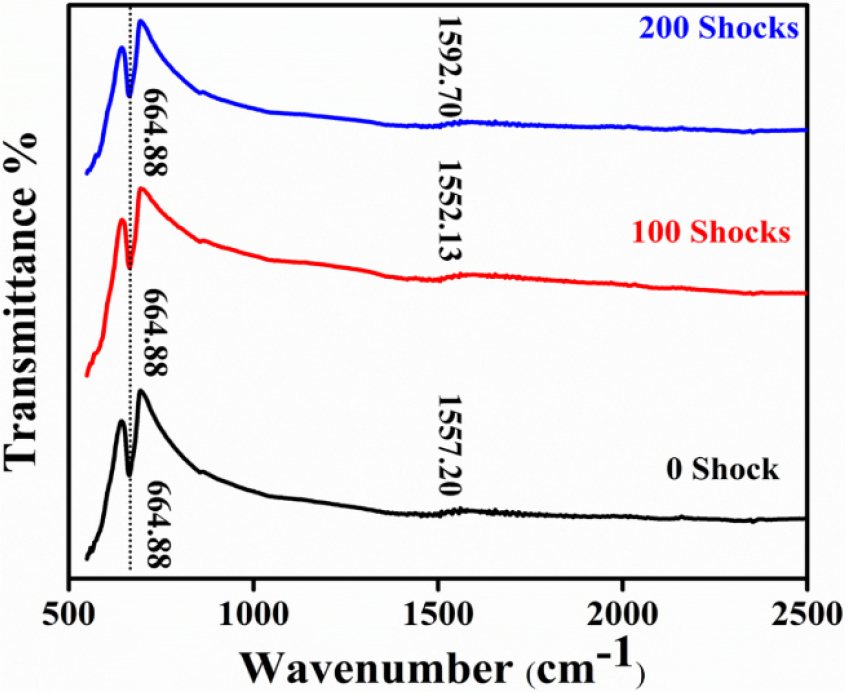
3.4 FTIR spectra analysis
In order to determine the functional groups and the bonds that are present in a material, FTIR spectra are utilized. It helps to confirm the structural integration or modifications for the LaCoO3/rGO composites. The FTIR spectra measured for the control and shocked samples in the range of 500-4000 cm-1 are shown in Fig. 6(a). Here, LaCoO3 and rGO are confirmed to coexist in all composites by FTIR, with rGO, C=C stretching in the 1520-1590 cm-1 range and Co-O stretching around ~540-560 cm-1. Changes in the rGO bonding environment are indicated by the systematic shift of the C=C band with increased shock wave; LCO/rGO 100 shocks exhibits more disorder, but, LCO/rGO 200 shocks shows improved graphitic characteristics. These FTIR observations, when combined with the Raman data, indicate that a mild shock wave maintains the oxide-carbon interface, whereas a higher shock increases contact and strain, which may change the electrical and catalytic properties.
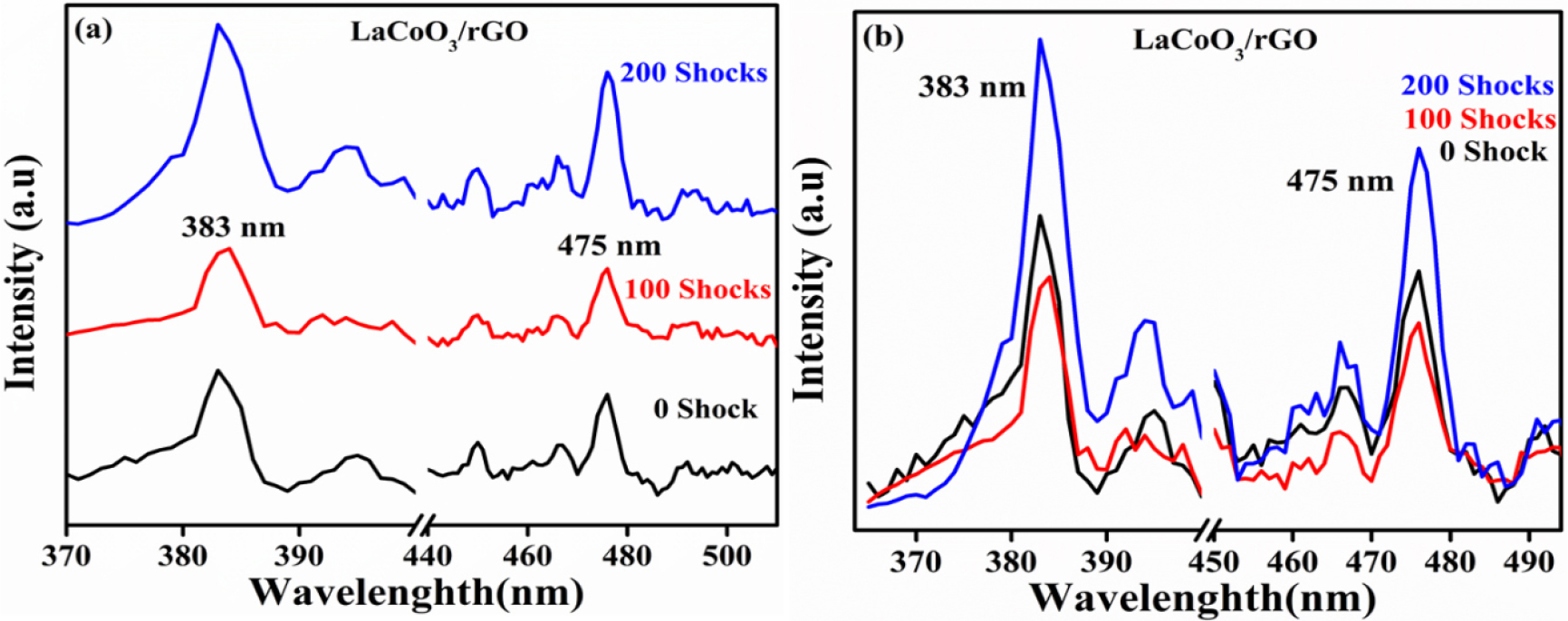
3.5 PL spectroscopy analysis
Photoluminescence spectroscopy (PL) were employed for control and shocked samples to investigate the recombination of change carriers within material. The PL spectra of control and shocked samples provide the charge separation efficiency, band gap transitions and defect states. In Fig. 7(a) and (b) show that, the two main emission bands of LaCoO3/rGO are a lower-energy, defect-related band near ~475 nm and a higher-energy band near ~383 nm. The PL changes with shock level show that the stronger shock (La-RGO-200) shifts the balance toward the 383 nm band (which is consistent with the formation of more shallow radiative defects, increased lattice strain/structural disorder, or altered band-to-band recombination). In contrast, the moderate shock (La-RGO-100) creates or activates a high density of radiative mid-gap defect centres (likely oxygen vacancies and/or an increased population of Co2+-related states). Overall, these trends suggest that shock treatment tunes the defect chemistry and the electronic coupling between LaCoO3 and rGO, alternately increasing radiative centers or promoting non-radiative charge transfer, depending on the number of shock pulses.
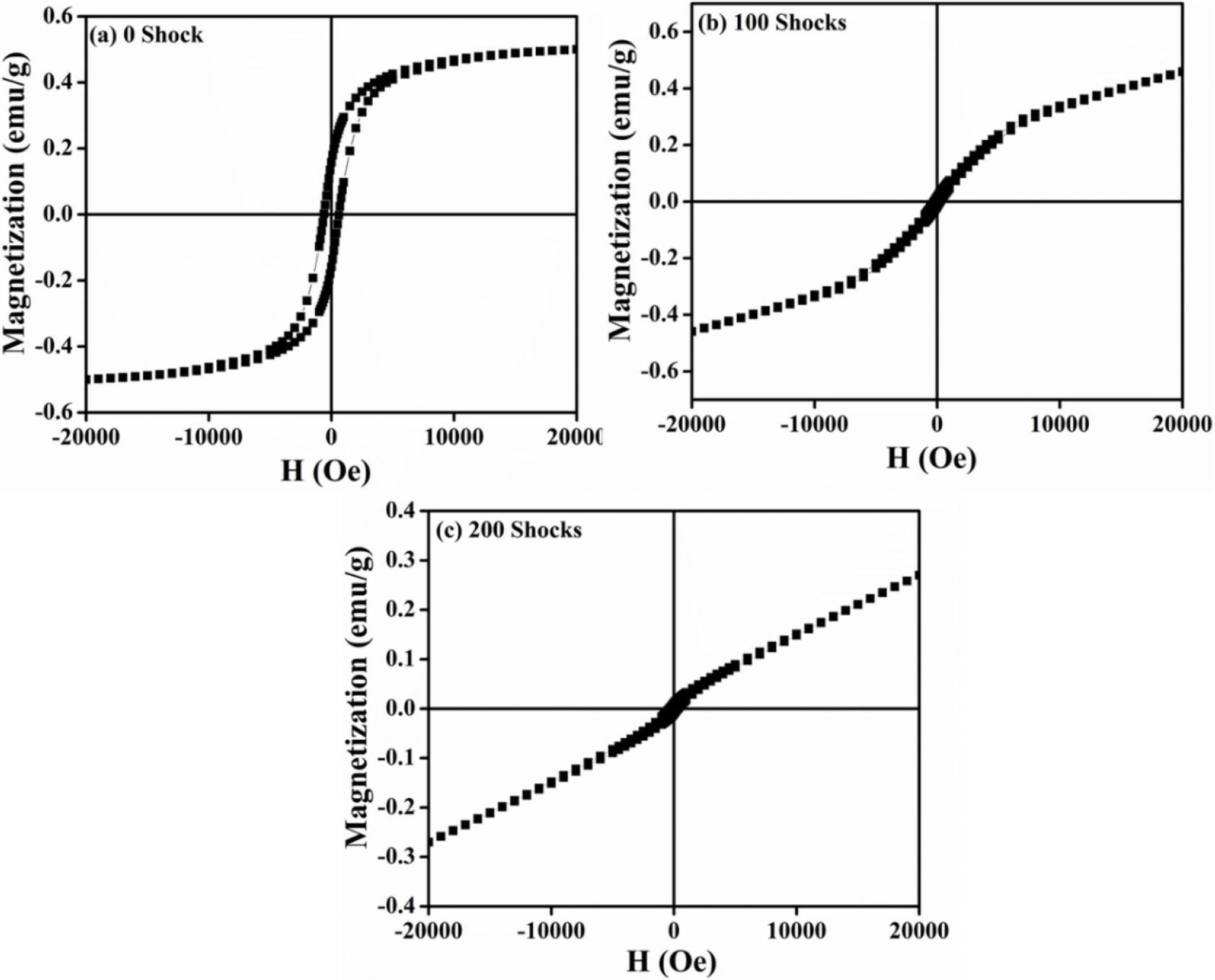
3.6 Isothermal magnetization analysis
The isothermal magnetization curve of the LaCoO3/rGO composite is shown in Fig. 8, in the magnetic field range of -2T to +2T. The magnetic response of LaCoO3 composites containing reduced graphene oxide (RGO) exhibits unique behavior. At 0 shock, LCO/rGO shows the strongest ferromagnetic-like behavior with clear saturation at high fields, indicating strong magnetic ordering due to interactions between LaCoO3 nanoparticles and RGO [16]. At 100 shocks, LCO/rGO shows moderate ferromagnetism with lower saturation magnetization, suggesting partial disruption of magnetic ordering by the shock wave. Interestingly, LCO/rGO-200 shows a mostly linear, paramagnetic-like response, suggesting that higher shock weakens Co-O-Co interactions, possibly due to structural distortion, spin-state changes, or reduced particle connectivity. The transition from S-shaped to linear curves highlights the suppression of ferromagnetism with increased shock treatment. Overall, the saturation magnetization falls from 0 shock to La-100 to La-200, and all samples show low coercivity, showing soft magnetic behaviour. Further, the changes in the S-shaped soft ferromagnetic to paramagnetic response under shocked conditions are due to spin canting induced by shock waves [32].
4. Conclusions
The nanocomposite of LCO/rGO was investigated under shocked conditions, such as control, 100, and 200 shocks. The material of LaCoO3 was prepared through hydrothermal techniques, and rGO was added through the ball milling method. For the shock wave treatment, the semi-automatic shock tube was used. The powder XRD confirms that the materials are hexagonal structures, and no extra peaks are observed. Under shocked conditions, a minor shift towards a lower angle is observed due to lattice expansion under shocked conditions. The morphology of the control and shocked materials was investigated through FESEM; it shows spherical particles, and the size of the particles slightly decreased due to shocks. Further, Raman and FTIR analyses show that shock intensity influences structural disorder, defects, and interfacial strain in LA-200, with mild shocks preserving the oxide-carbon interface and stronger shocks inducing disorder. PL analysis reveals that these shocks also modulate defect chemistry and electronic interactions in LaCoO3/rGO, affecting radiative and non-radiative charge transfer. Under shocked conditions, the isothermal magnetization curve changes its soft ferromagnetic to paramagnetic nature due to spin canting induced by pressure and temperature. Moreover, the lack of changes in structural, vibrational, and optical properties confirms that the materials possess robust characteristics. Hence, the material is used for aerospace and defense applications.